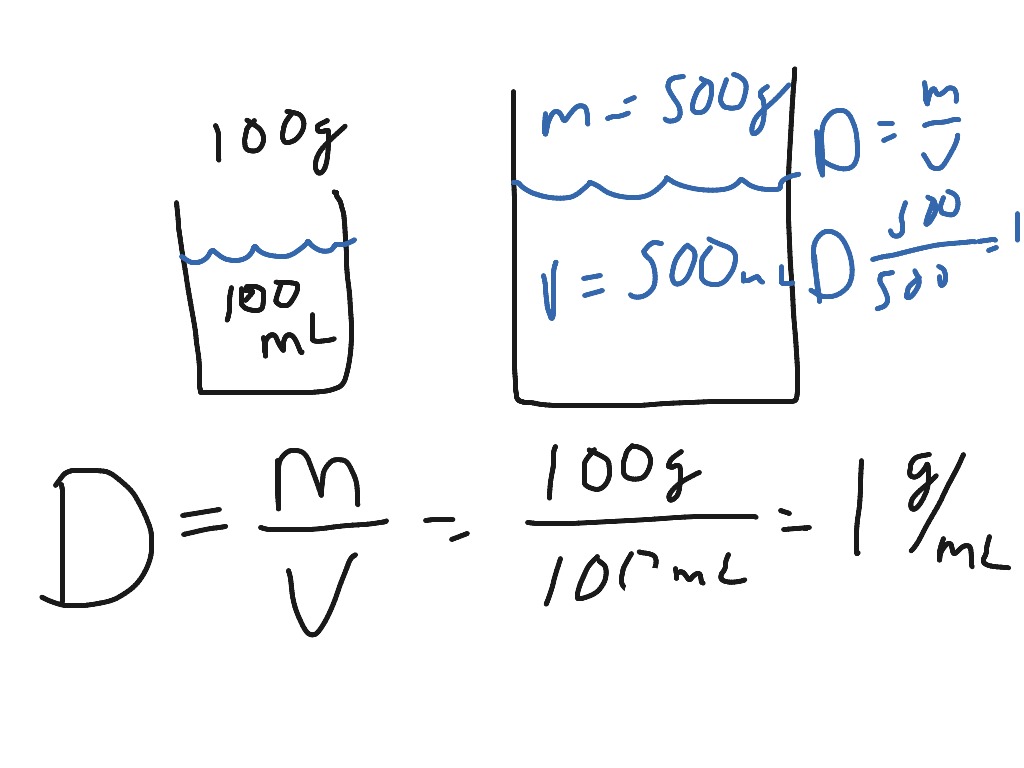
Similarly, a decrease in solute concentration decreases the specific gravity of urine, which indicates medical problems like renal failure. An increase in the specific gravity of urine indicates that it is due to an increase in the solutes caused by dehydration, diarrhea, or infection. Both high and low density or specific gravity than the normal range of urine indicates medical problems. Both the blood and urine have dissolved substances in water that increase the density from that of pure water. 
The density of urine varies in the range of 1.0050 g/mL to 1.030 g/mL. For example, the density of whole blood for humans is ~1.060 g/mL. When substances dissolve in water, the density of the solution is usually different from pure water. The density of water is 1.0 g/mL at room temperature, so the specific gravity is equal to the density of the object expressed without a unit. Therefore, the specific gravity is a unitless number. We thank the University of Michigan and the NOAA for assistance with this calculator and information.\nonumber You just need to measure the salinity, temperature and pressure to be able to find density. Density is usually calculated using a standard equation. If you wanted to measure the density of ocean water but did not have the expensive equipment to do it directly, you would have to collect a sample of sea water and bring it back to a controlled environment to be measured. The density of ocean water is rarely measured directly. Hence, ocean water moves laterally along the layers with the same density. That surface level movement is to some degree vertical and horizontal, perhaps swirling, but within the same general density layer. Circulation in the depths of the ocean is horizontal, a result of deviations in density in constrast to surface circulation which is a product of surface winds. The deep ocean is layered with the most dense water on bottom and the lightest water on top. So, the density of ocean water increases somewhat proportionately as you go to or toward the bottom of the ocean. The temperature of the ocean decreases significantly as you go to the bottom of the ocean. Hence, a layer of water (pycnocline) with higher salinity can actual float on top of water with lower salinity if the layer with higher salinity is quite a bit warmer than the lower salinity layer. However, temperature has a greater effect on the density of water than does salinity. Given two layers of water with the same salinity, the warmer water will float on top of the colder water. Less dense water floats on top of more dense water. Question 2: A solution of water and salt contains 25 grams of salt in 250 mL of water. Answer 1: The sugar cube has a density of 1.4 grams/cm 3. Increasing salinity also increases the density of sea water. Step 2: Plug your variables into the density formula. So, the colder the water, the more dense it is. Ocean water gets more dense as temperature goes down. The first is the temperature of the water and the other us the salinity of the water. There are two main factors that make ocean water more or less dense than about 1027 kg/m 3. Density of ocean water at the sea surface is about 1027 kg/m 3. Ocean water is more dense because of the salt in it. The density of pure water is 1000 kg/m 3. There are exceptions however, such as water's density increasing between 0C and 4C. Increases in temperature tend to decrease density since the volume will generally increase. An increase in pressure decreases volume, and always increases density. This is applicable for any fluid that is primarily water based. However, when regarding gases, density is largely affected by temperature and pressure. Then click on the Calculate button and the water density is calculated and displayed, based on your input data. In our calculator, enter the temperature in degrees C or F (click anywhere outside of any of the fields for the conversion), and the salinity in mg/L or the equivalent of PPM. Use this calculator to account for pressure. 
Pressure is a large factor in water density in the ocean. This script determines the density of water is a function of both temperature and salinity pressure is assumed at the surface since this is for non-depth related samples. This calculator requires the use of Javascript enabled and capable browsers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
