|
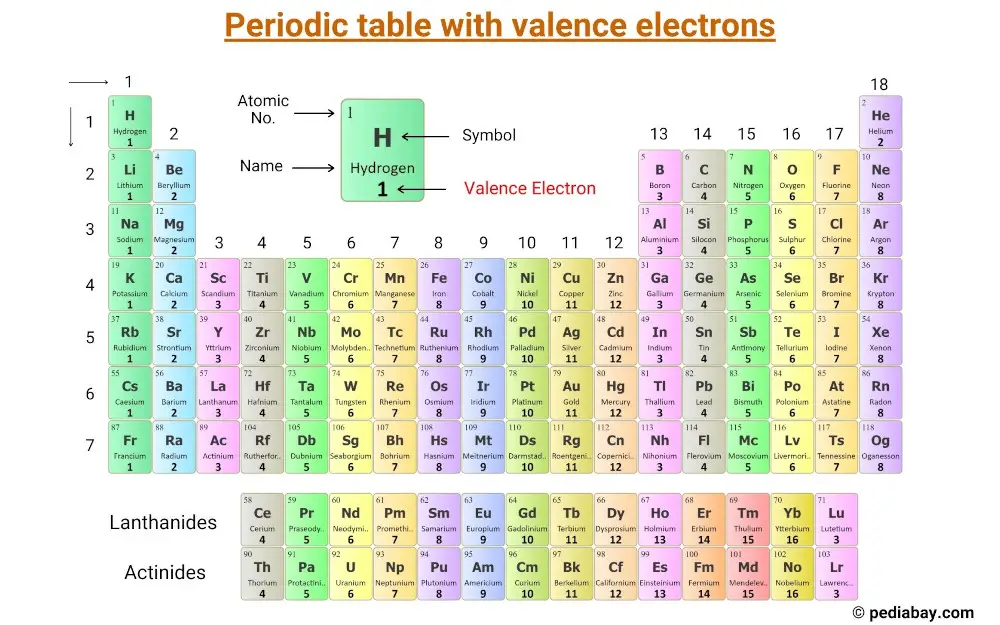
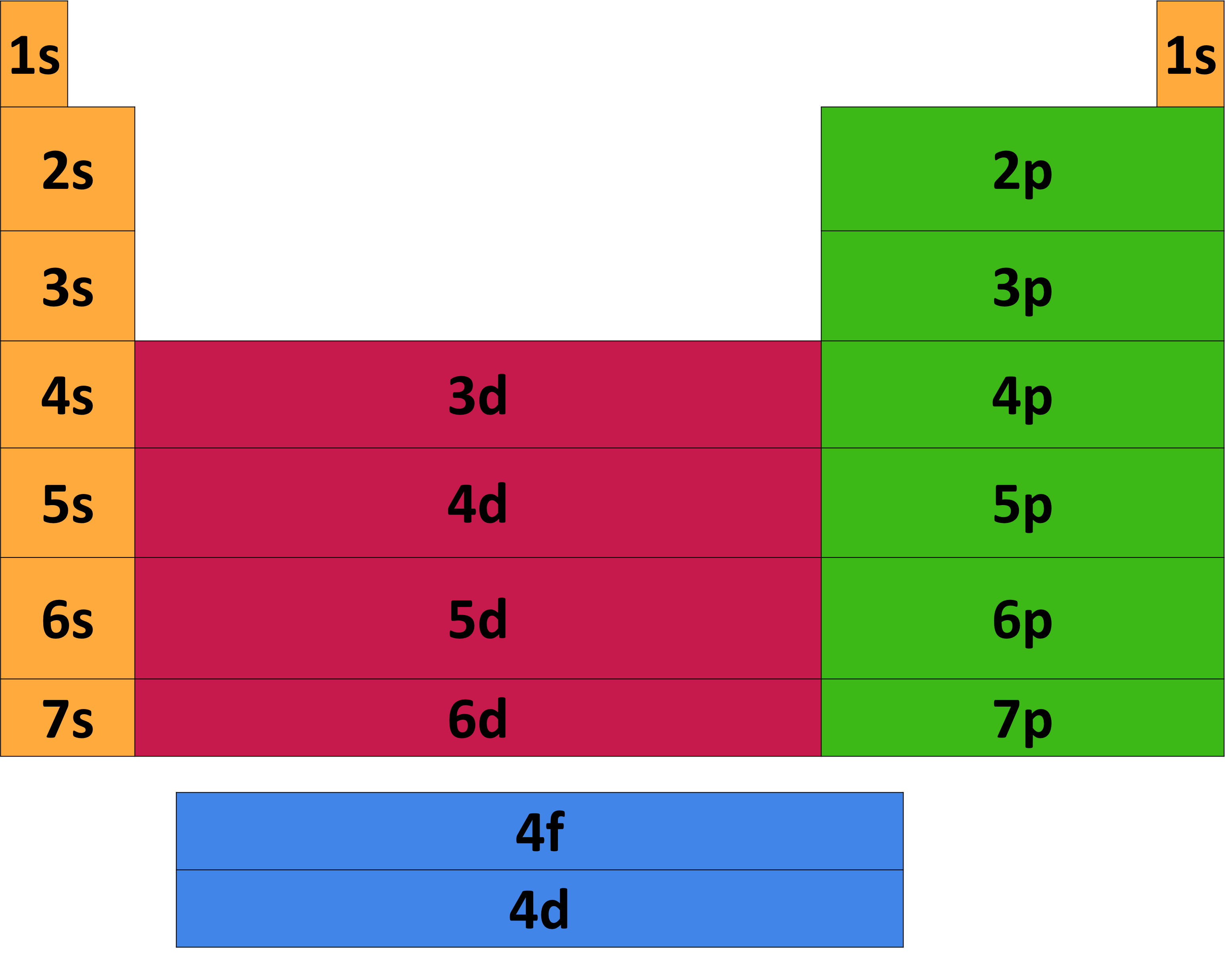
The concept of electronic configuration has replaced the older concept of valency and valence electrons. In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. Study with Quizlet and memorize flashcards containing terms like They have the same valence electron configuration (sp), The energy level of an atoms. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. When we reach neon, with Z = 10, we have filled the 2 p subshell, giving a 1 s 22 s 22 p 6 electron configuration:Īs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. Similarly, fluorine has the electron configuration 1 s 22 s 22 p 5: Fluorine has one electron pair in the 1 s orbital, one electron pair in the 2 s orbital, and 2 electrons pairs with one unpaired electron in the 2 p orbital. Oxygen has one electron pair in the 1 s orbital, one electron pair in the 2 s pair, and one electron pair, and 2 unpaired electrons in the 2 p orbitals. 1: Periodic table by Dmitri Mendeleev, 1871. After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleev's periodic table. Because all the 2 p orbitals are degenerate, it doesn’t matter which one has the pair of electrons. As a gas or vapor, the halogens all had a pungent odor. 
One electron must be paired with another in one of the 2 p orbitals, which gives us two unpaired electrons and a 1 s 22 s 22 p 4 electron configuration. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice. Include examples and state the trend in valence electrons within a group on the periodic table.\): Copy and Paste Caption here. These two elements make up the first row of the periodic table (Figure 9.7.2 9.7. For example, all the alkali metals have 'isoelectronic' valence shell electron configurations. Their electron configurations are 1 s 1 and 1 s 2, respectively with He, the n 1 shell is filled. The ground state configuration is the lowest energy configuration, and the ground state electron configuration affects the behavior of atoms and results in the elemental structure of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
